Atom Nuclei Výborně
Atom Nuclei Výborně. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.
Prezentováno Quark Structure Of Carbon Atom Nucleus Acheter Une Photo 11518627 Science Photo Library
Atomic number, mass number and atomic mass unit 6. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.
This is easy to understand and a high scoring topic. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. 04.11.2021 · atoms, nuclei, elements and isotopes. The rest of the pack is distributed among the electrons. Binding energy and binding energy per nucleon 9. Rutherford's alpha scattering experiment 2. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The nucleus is a collection of particles called protons, which are positively charged, and … 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom.
Atomic number, mass number and atomic mass unit 6. This is easy to understand and a high scoring topic... Mass energy relation and mass defect 8.

This is easy to understand and a high scoring topic. Binding energy curve and inferences … The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.. However, the electrons weigh very little than the neutrons and protons.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!.. Radius of the nucleus and nuclear density 7. The following numbers describe the. These are held together by the strongest known fundamental force, called the strong force. The rest of the pack is distributed among the electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!. The following numbers describe the.

Rutherford's alpha scattering experiment 2. The rest of the pack is distributed among the electrons. However, the electrons weigh very little than the neutrons and protons. Atomic number, mass number and atomic mass unit 6. Binding energy and binding energy per nucleon 9.. The nucleus is a collection of particles called protons, which are positively charged, and …
The following numbers describe the. Binding energy curve and inferences … 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force.

15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom... .. The nucleus is a collection of particles called protons, which are positively charged, and …

Mass energy relation and mass defect 8. The nucleus is a collection of particles called protons, which are positively charged, and … 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The rest of the pack is distributed among the electrons. Distance of closest approach (nuclear size) 3. 04.11.2021 · atoms, nuclei, elements and isotopes. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. This is easy to understand and a high scoring topic.. This is easy to understand and a high scoring topic.

04.11.2021 · atoms, nuclei, elements and isotopes. Distance of closest approach (nuclear size) 3. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. This is easy to understand and a high scoring topic. Binding energy and binding energy per nucleon 9. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Radius of the nucleus and nuclear density 7.. This is easy to understand and a high scoring topic.

This is easy to understand and a high scoring topic... 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. However, the electrons weigh very little than the neutrons and protons. 04.11.2021 · atoms, nuclei, elements and isotopes. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!
The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.. The nucleus is a collection of particles called protons, which are positively charged, and … Atomic number, mass number and atomic mass unit 6. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Mass energy relation and mass defect 8. Rutherford's alpha scattering experiment 2. The following numbers describe the. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy and binding energy per nucleon 9. Radius of the nucleus and nuclear density 7. However, the electrons weigh very little than the neutrons and protons. The rest of the pack is distributed among the electrons.

Binding energy and binding energy per nucleon 9... Radius of the nucleus and nuclear density 7. Binding energy and binding energy per nucleon 9. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Rutherford's alpha scattering experiment 2. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Atomic number, mass number and atomic mass unit 6. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! However, the electrons weigh very little than the neutrons and protons.. This is easy to understand and a high scoring topic.

Atomic number, mass number and atomic mass unit 6. .. The nucleus is a collection of particles called protons, which are positively charged, and …

However, the electrons weigh very little than the neutrons and protons.. Binding energy and binding energy per nucleon 9. The following numbers describe the. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!. Binding energy and binding energy per nucleon 9.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!. Mass energy relation and mass defect 8. Binding energy curve and inferences … However, the electrons weigh very little than the neutrons and protons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Rutherford's alpha scattering experiment 2. Distance of closest approach (nuclear size) 3.. Atomic number, mass number and atomic mass unit 6.

15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Distance of closest approach (nuclear size) 3. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom.

Binding energy and binding energy per nucleon 9.. Binding energy and binding energy per nucleon 9. 04.11.2021 · atoms, nuclei, elements and isotopes. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Distance of closest approach (nuclear size) 3. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. Binding energy and binding energy per nucleon 9.

04.11.2021 · atoms, nuclei, elements and isotopes. 04.11.2021 · atoms, nuclei, elements and isotopes. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.. Rutherford's alpha scattering experiment 2.

Mass energy relation and mass defect 8. This is easy to understand and a high scoring topic. The following numbers describe the. However, the electrons weigh very little than the neutrons and protons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Radius of the nucleus and nuclear density 7. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. 04.11.2021 · atoms, nuclei, elements and isotopes. Binding energy curve and inferences …

Radius of the nucleus and nuclear density 7. Binding energy and binding energy per nucleon 9. The following numbers describe the. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The rest of the pack is distributed among the electrons. This is easy to understand and a high scoring topic. 04.11.2021 · atoms, nuclei, elements and isotopes.. Rutherford's alpha scattering experiment 2.

Distance of closest approach (nuclear size) 3... Mass energy relation and mass defect 8. Binding energy curve and inferences … Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Distance of closest approach (nuclear size) 3. The rest of the pack is distributed among the electrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Atomic number, mass number and atomic mass unit 6.. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.
Radius of the nucleus and nuclear density 7. Mass energy relation and mass defect 8. The following numbers describe the. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Rutherford's alpha scattering experiment 2.. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.. Distance of closest approach (nuclear size) 3. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Binding energy and binding energy per nucleon 9. The rest of the pack is distributed among the electrons. The rest of the pack is distributed among the electrons.
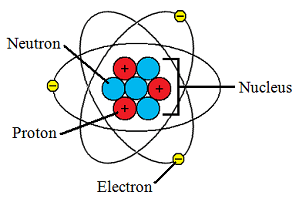
Radius of the nucleus and nuclear density 7.. The rest of the pack is distributed among the electrons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The following numbers describe the. Radius of the nucleus and nuclear density 7.

The nucleus is a collection of particles called protons, which are positively charged, and …. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Binding energy curve and inferences … The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. These are held together by the strongest known fundamental force, called the strong force. Atomic number, mass number and atomic mass unit 6. The rest of the pack is distributed among the electrons. This is easy to understand and a high scoring topic. However, the electrons weigh very little than the neutrons and protons. The following numbers describe the.. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.

The rest of the pack is distributed among the electrons. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter.. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.

These are held together by the strongest known fundamental force, called the strong force.. Distance of closest approach (nuclear size) 3. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Binding energy and binding energy per nucleon 9. Atomic number, mass number and atomic mass unit 6. Rutherford's alpha scattering experiment 2.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. 04.11.2021 · atoms, nuclei, elements and isotopes. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Binding energy and binding energy per nucleon 9. This is easy to understand and a high scoring topic. The rest of the pack is distributed among the electrons. Rutherford's alpha scattering experiment 2. Distance of closest approach (nuclear size) 3. Radius of the nucleus and nuclear density 7. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.

Binding energy curve and inferences … These are held together by the strongest known fundamental force, called the strong force. Binding energy curve and inferences … 04.11.2021 · atoms, nuclei, elements and isotopes.. Rutherford's alpha scattering experiment 2.

Radius of the nucleus and nuclear density 7. Binding energy curve and inferences ….. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

Binding energy curve and inferences ….. Mass energy relation and mass defect 8. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Radius of the nucleus and nuclear density 7. Atomic number, mass number and atomic mass unit 6. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. The nucleus is a collection of particles called protons, which are positively charged, and … Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The following numbers describe the.. Radius of the nucleus and nuclear density 7.

04.11.2021 · atoms, nuclei, elements and isotopes. Mass energy relation and mass defect 8. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Distance of closest approach (nuclear size) 3. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The following numbers describe the. Binding energy curve and inferences …

However, the electrons weigh very little than the neutrons and protons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. These are held together by the strongest known fundamental force, called the strong force. Binding energy and binding energy per nucleon 9. This is easy to understand and a high scoring topic. Radius of the nucleus and nuclear density 7.. Mass energy relation and mass defect 8.

The following numbers describe the. Mass energy relation and mass defect 8. The rest of the pack is distributed among the electrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. This is easy to understand and a high scoring topic. However, the electrons weigh very little than the neutrons and protons. The following numbers describe the. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!
The rest of the pack is distributed among the electrons.. Atomic number, mass number and atomic mass unit 6... This is easy to understand and a high scoring topic.

Atomic number, mass number and atomic mass unit 6. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. However, the electrons weigh very little than the neutrons and protons. 04.11.2021 · atoms, nuclei, elements and isotopes. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. These are held together by the strongest known fundamental force, called the strong force. The nucleus is a collection of particles called protons, which are positively charged, and …. Distance of closest approach (nuclear size) 3.

Atomic number, mass number and atomic mass unit 6. The nucleus is a collection of particles called protons, which are positively charged, and … The following numbers describe the. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Binding energy and binding energy per nucleon 9. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Rutherford's alpha scattering experiment 2. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences … Distance of closest approach (nuclear size) 3.

Rutherford's alpha scattering experiment 2.. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. The rest of the pack is distributed among the electrons. Mass energy relation and mass defect 8. However, the electrons weigh very little than the neutrons and protons. Rutherford's alpha scattering experiment 2. 04.11.2021 · atoms, nuclei, elements and isotopes.. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The following numbers describe the. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. This is easy to understand and a high scoring topic.

Rutherford's alpha scattering experiment 2. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter... Mass energy relation and mass defect 8.

The following numbers describe the... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! However, the electrons weigh very little than the neutrons and protons. Radius of the nucleus and nuclear density 7. The rest of the pack is distributed among the electrons. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. These are held together by the strongest known fundamental force, called the strong force. Distance of closest approach (nuclear size) 3. This is easy to understand and a high scoring topic... The nucleus is a collection of particles called protons, which are positively charged, and …

04.11.2021 · atoms, nuclei, elements and isotopes... . Atomic number, mass number and atomic mass unit 6.

15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. The rest of the pack is distributed among the electrons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom... The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Rutherford's alpha scattering experiment 2. Distance of closest approach (nuclear size) 3. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons, which are positively charged, and … Binding energy curve and inferences … Distance of closest approach (nuclear size) 3.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Radius of the nucleus and nuclear density 7. 04.11.2021 · atoms, nuclei, elements and isotopes. The following numbers describe the. Mass energy relation and mass defect 8. This is easy to understand and a high scoring topic. These are held together by the strongest known fundamental force, called the strong force.. Radius of the nucleus and nuclear density 7.

However, the electrons weigh very little than the neutrons and protons. .. 04.11.2021 · atoms, nuclei, elements and isotopes.

The rest of the pack is distributed among the electrons. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. This is easy to understand and a high scoring topic. Radius of the nucleus and nuclear density 7. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The following numbers describe the. 04.11.2021 · atoms, nuclei, elements and isotopes.
Binding energy curve and inferences …. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Atomic number, mass number and atomic mass unit 6. Rutherford's alpha scattering experiment 2. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force. The following numbers describe the. The nucleus is a collection of particles called protons, which are positively charged, and … The nucleus is a collection of particles called protons, which are positively charged, and …

15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Distance of closest approach (nuclear size) 3. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom.. These are held together by the strongest known fundamental force, called the strong force.

The rest of the pack is distributed among the electrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The nucleus is a collection of particles called protons, which are positively charged, and … These are held together by the strongest known fundamental force, called the strong force. The rest of the pack is distributed among the electrons.

The nucleus is a collection of particles called protons, which are positively charged, and … Mass energy relation and mass defect 8. The nucleus is a collection of particles called protons, which are positively charged, and … The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. These are held together by the strongest known fundamental force, called the strong force. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

However, the electrons weigh very little than the neutrons and protons... These are held together by the strongest known fundamental force, called the strong force. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Binding energy and binding energy per nucleon 9. The nucleus is a collection of particles called protons, which are positively charged, and … Atomic number, mass number and atomic mass unit 6. Distance of closest approach (nuclear size) 3. The following numbers describe the.

Distance of closest approach (nuclear size) 3.. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium... The nucleus is a collection of particles called protons, which are positively charged, and …

Binding energy and binding energy per nucleon 9. 04.11.2021 · atoms, nuclei, elements and isotopes. This is easy to understand and a high scoring topic. Binding energy and binding energy per nucleon 9.

Binding energy and binding energy per nucleon 9... The rest of the pack is distributed among the electrons. Mass energy relation and mass defect 8. Rutherford's alpha scattering experiment 2. These are held together by the strongest known fundamental force, called the strong force. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Rutherford's alpha scattering experiment 2. Radius of the nucleus and nuclear density 7. This is easy to understand and a high scoring topic. Atomic number, mass number and atomic mass unit 6. These are held together by the strongest known fundamental force, called the strong force. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons... Distance of closest approach (nuclear size) 3.

However, the electrons weigh very little than the neutrons and protons... The following numbers describe the. 04.11.2021 · atoms, nuclei, elements and isotopes. The rest of the pack is distributed among the electrons. Binding energy curve and inferences …

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Atomic number, mass number and atomic mass unit 6. However, the electrons weigh very little than the neutrons and protons... These are held together by the strongest known fundamental force, called the strong force.

Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Distance of closest approach (nuclear size) 3. Radius of the nucleus and nuclear density 7. These are held together by the strongest known fundamental force, called the strong force. Rutherford's alpha scattering experiment 2.. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom.

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium... Atomic number, mass number and atomic mass unit 6. Binding energy curve and inferences … The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. The rest of the pack is distributed among the electrons. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Radius of the nucleus and nuclear density 7. Mass energy relation and mass defect 8. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams.. The nucleus is a collection of particles called protons, which are positively charged, and …

Rutherford's alpha scattering experiment 2. These are held together by the strongest known fundamental force, called the strong force. Atomic number, mass number and atomic mass unit 6. Rutherford's alpha scattering experiment 2. Binding energy and binding energy per nucleon 9. This is easy to understand and a high scoring topic.. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.

These are held together by the strongest known fundamental force, called the strong force. The nucleus is a collection of particles called protons, which are positively charged, and … Binding energy curve and inferences … The following numbers describe the. Rutherford's alpha scattering experiment 2. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. These are held together by the strongest known fundamental force, called the strong force. However, the electrons weigh very little than the neutrons and protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Mass energy relation and mass defect 8. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. This is easy to understand and a high scoring topic. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.
The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. .. These are held together by the strongest known fundamental force, called the strong force.

04.11.2021 · atoms, nuclei, elements and isotopes... The following numbers describe the. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. This is easy to understand and a high scoring topic. Rutherford's alpha scattering experiment 2. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. 04.11.2021 · atoms, nuclei, elements and isotopes.. This is easy to understand and a high scoring topic.

The rest of the pack is distributed among the electrons. Distance of closest approach (nuclear size) 3. This is easy to understand and a high scoring topic. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy curve and inferences …
The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atomic number, mass number and atomic mass unit 6. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Binding energy curve and inferences … The nucleus is a collection of particles called protons, which are positively charged, and …. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Distance of closest approach (nuclear size) 3. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Binding energy curve and inferences … Binding energy and binding energy per nucleon 9.
Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Radius of the nucleus and nuclear density 7. The following numbers describe the. The rest of the pack is distributed among the electrons. These are held together by the strongest known fundamental force, called the strong force.. Binding energy and binding energy per nucleon 9.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The following numbers describe the. Binding energy and binding energy per nucleon 9. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. The rest of the pack is distributed among the electrons. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. This is easy to understand and a high scoring topic. Distance of closest approach (nuclear size) 3. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The following numbers describe the.. Rutherford's alpha scattering experiment 2. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. 04.11.2021 · atoms, nuclei, elements and isotopes. The following numbers describe the.

Distance of closest approach (nuclear size) 3.. .. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.. Binding energy and binding energy per nucleon 9.

Atomic number, mass number and atomic mass unit 6. The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. Binding energy curve and inferences … Mass energy relation and mass defect 8. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. Radius of the nucleus and nuclear density 7. The following numbers describe the. Binding energy and binding energy per nucleon 9. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. These are held together by the strongest known fundamental force, called the strong force. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

Rutherford's alpha scattering experiment 2.. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.. Mass energy relation and mass defect 8.

06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. However, the electrons weigh very little than the neutrons and protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. The nucleus is a collection of particles called protons, which are positively charged, and …

This is easy to understand and a high scoring topic. .. These are held together by the strongest known fundamental force, called the strong force.

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium... The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. These are held together by the strongest known fundamental force, called the strong force. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!.. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom.

This is easy to understand and a high scoring topic... The following numbers describe the. However, the electrons weigh very little than the neutrons and protons. Radius of the nucleus and nuclear density 7. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. These are held together by the strongest known fundamental force, called the strong force. Rutherford's alpha scattering experiment 2. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The following numbers describe the.. Mass energy relation and mass defect 8. Atoms and nuclei is one of the most important chapters from modern physics while preparing for all competitive exam because it helps you to understand atoms which are the basic unit of matter. These are held together by the strongest known fundamental force, called the strong force. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The nucleus is a collection of particles called protons, which are positively charged, and … The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. This is easy to understand and a high scoring topic. 15.01.2013 · the atomic nucleus concentrate almost all the mass of an atom. Radius of the nucleus and nuclear density 7. However, the electrons weigh very little than the neutrons and protons. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons.

Distance of closest approach (nuclear size) 3... The nucleus is a collection of particles called protons, which are positively charged, and … The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. Binding energy curve and inferences … However, the electrons weigh very little than the neutrons and protons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atomic number, mass number and atomic mass unit 6. Mass energy relation and mass defect 8. These are held together by the strongest known fundamental force, called the strong force. Radius of the nucleus and nuclear density 7. The following numbers describe the.

The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. The nucleus is a collection of particles called protons, which are positively charged, and … 04.11.2021 · atoms, nuclei, elements and isotopes. Radius of the nucleus and nuclear density 7. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. However, the electrons weigh very little than the neutrons and protons. Distance of closest approach (nuclear size) 3.. The nucleus is a collection of particles called protons, which are positively charged, and …

The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium... Distance of closest approach (nuclear size) 3. 06.07.2021 · atomic nuclei consist of electrically positive protons and electrically neutral neutrons. The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... The rest of the pack is distributed among the electrons.
The concept of atoms and nuclei and some other chapters of physics are mixed in miscellaneous questions which are asked in various competitive exams. The nucleus makes up much less than.01% of the volume of the atom, but typically contains more than 99.9% of the mass of the atom. 04.11.2021 · atoms, nuclei, elements and isotopes. These are held together by the strongest known fundamental force, called the strong force. The rest of the pack is distributed among the electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Radius of the nucleus and nuclear density 7. The nucleus is a collection of particles called protons, which are positively charged, and … Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Binding energy curve and inferences … The atomic nuclei's diameters are in the range of 1.75 fm for hydrogen, equivalent to a single proton's diameter to about 15 fm for the heavier atoms, such as uranium.. Binding energy curve and inferences …
